AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog

(b) Use the balanced equation to determine the mole ratio MgO:Mg mass MgO = moles(MgO) × molar mass(MgO).(c) Use the mole ratio to calculate moles O 2 (b) Use the balanced chemical equation to determine the mole ratio O 2:Mg (a) Calculate moles Mg = mass(Mg) ÷ molar mass(Mg) mass O 2 = moles(O 2) × molar mass(O 2).It is possible to calculate the mass of each reactant and product using the mole ratio (stoichiometric ratio) from the balanced chemical equation and the mathematical equation moles = mass ÷ molar massįor the balanced chemical equation shown below: Take the test now! Chemical Reactions and Masses of Reactants and Products 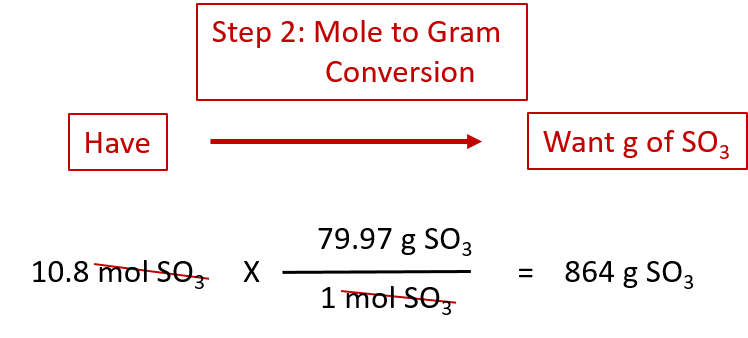
The mole ratio is the stoichiometric ratio of reactants and products and is the ratio of the stoichiometric coefficients for reactants and products found in the balanced chemical equation. No ads = no money for us = no free stuff for you! Mole Ratio (stoichiometric ratio) The mole ratio (stoichiometric ratio) can be used to calculate the mass of reactants and products in a chemical reaction.The ratio of moles of each reactant and product in a reaction is known as the mole ratio (or stoichiometric ratio).⚛ The ratio of the moles of each reactant and product. ⚛ The ratio of the number of molecules of each type reacting and produced. A balanced chemical equation can tell us:.You need to become an AUS-e-TUTE Member! Calculating Moles and Mass in Chemical Reactions Using Mole Ratios (stoichoimetric ratios) Chemistry Tutorial Key Concepts Want chemistry games, drills, tests and more? Calculating Moles and Mass in Reactions Chemistry Tutorial More Free Tutorials Become a Member Members Log‐in Contact Us
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
